Introduction
In biotechnology and medicine, achieving efficient drug delivery across biological membranes while preserving cell viability remains a major challenge.
In this context, sonoporation is emerging as an innovative ultrasound-based therapy for targeted drug delivery. By using ultrasound to induce transient membrane permeability, this technique enables enhanced intracellular delivery of therapeutic agents, including small molecules, biologics, and nanoparticles.
However, the complexity and stochastic nature of acoustic cavitation make it difficult to achieve precise and reproducible control of these effects. Mastering these parameters is essential for improving treatment efficiency and ensuring safety in both biomedical research and clinical applications.
In this article, we explore the principles, applications, and key advantages of sonoporation, as well as the ultrasound technologies we develop to optimize control, reproducibility, and performance in advanced drug delivery systems.
What is sonoporation?
Sonoporation is an innovative technique, primarily used in preclinical research and gene therapy, aimed at improving the efficiency of drug delivery. It relies on the use of ultrasound to enhance the uptake of therapeutic agents—such as drugs, plasmids, DNA, RNA, or nanoparticles—into biological cells.
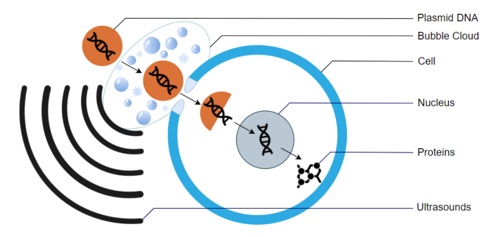
This process temporarily increases cell membrane permeability, allowing therapeutic agents to enter cells more efficiently. This permeabilization is transient and reversible: the cell membrane returns to its initial state after ultrasound exposure, preserving cell viability.
In this context, sonoporation stands out as a highly promising targeted drug delivery approach, particularly for applications where cellular uptake represents a major barrier. It enables improved diffusion and absorption of therapeutic compounds into tissues or cells that are otherwise difficult to access.
Sonoporation is currently being widely studied in preclinical research, especially to enhance the efficiency of targeted delivery strategies, and is attracting growing interest in the development of new non-invasive therapeutic approaches based on ultrasound technology.
How Sonoporation Works
The mechanism of sonoporation is primarily based on the mechanical effects of ultrasound in a liquid medium.
Under an ultrasonic field, microbubbles undergo oscillations. Depending on the acoustic conditions, these oscillations may remain moderate (stable cavitation) or become more intense (inertial cavitation). These dynamics generate phenomena such as microstreaming, microjet formation, and shock waves, leading to localized mechanical stresses on cell membranes.
These mechanical effects result in:
🔹 Transient deformation of the cell membrane
The membrane is subjected to cyclic pressure variations, causing temporary deformation. This structural modification increases its permeability to therapeutic molecules.
🔹 Microstreaming-induced fluid motion
Oscillating microbubbles generate localized fluid movements, creating shear forces at the cell surface. These forces enhance the transport of molecules toward the membrane.
🔹 Formation of transient pores in the cell membrane
As a combined effect of these mechanisms, temporary openings form in the membrane. These pores allow therapeutic agents to enter the cell before resealing after ultrasound exposure.
All these phenomena strongly depend on acoustic parameters (frequency, pressure, duration) as well as on the properties of the medium, particularly the presence and concentration of microbubbles. Controlling these parameters is essential to achieve an optimal balance between drug delivery efficiency and cell safety.
Scientific Challenges of Sonoporation
Although the mechanisms of sonoporation are now better understood, achieving precise control remains a major challenge for researchers—particularly in terms of reliability and reproducibility.
This difficulty stems from the complexity of the underlying phenomena: biological mechanisms that are still only partially understood, acoustic cavitation that is highly sensitive to multiple parameters and inherently variable, and a strong dependence on experimental conditions. In addition, there is a delicate balance between efficiency and safety, as well as the lack of standardized protocols for robust clinical translation.
In this context, the challenge is no longer limited to understanding these mechanisms, but rather to controlling them in a precise and predictable way, despite their non-linear and partially stochastic nature.
This requires the development of advanced ultrasound systems capable of precisely managing emission parameters—such as frequency, power, and signal shape—while also monitoring induced effects in real time. Such control is essential to reduce variability and improve the reproducibility of results in drug delivery applications.
It is precisely within this framework of control and optimization of ultrasound-driven phenomena that the solutions developed by SinapTec are positioned.
Solutions Générateur Laboratoire SinapTec pour la sonoporation
In this context, and in response to the increasing demands for control and reproducibility in sonoporation, SinapTec develops modular ultrasound generators based on our NexTgen® platform, designed to be easily integrated and adapted to advanced biomedical protocols.
We prioritize a single-channel solution tailored for laboratory use, while also offering a multi-channel option suited to more complex structural requirements, ensuring optimal reproducibility of treatments in research environments.
Our generators provide full control over treatment conditions through advanced features:
- Precise control of ultrasound parameters (frequency, power, duration, and pulse sequences),
- Real-time measurement of power delivered to the transducer,
- Reduction of experimental variability to improve reproducibility,
- Cavitation level assessment through spectral analysis, including the use of appropriate hydrophones.

These technologies improve protocol reproducibility and enable better control of the underlying physical phenomena—an essential requirement for sonoporation and ultrasound-mediated drug delivery applications in laboratory settings.
Custom Ultrasound Solutions
Each sonoporation application involves specific constraints, depending on the targeted tissues, the therapeutic agents delivered, and the experimental conditions.
This is why we offer custom ultrasound solutions, tailored to your scientific or clinical objectives. We support you in:
- Ultrasound generator design,
- Definition of acoustic parameters,
- Transducer adaptation,
- Protocol optimization.
This approach enables the development of systems fully aligned with your needs, whether for fundamental research or preclinical validation.
Sonoporation: Applications in Medicine and Biopharmaceuticals
🔹 Gene Therapy
In gene therapy, sonoporation represents a promising alternative to viral vectors for the intracellular delivery of genetic material (plasmids, DNA, RNA).
It enhances the efficiency of ultrasound-mediated transfection while reducing the risks associated with biological vectors, such as immune responses or toxicity.
This ultrasound-based gene delivery approach is widely studied in preclinical research for targeted and controlled applications.
🔹 Neurology
In neurology, sonoporation enables the transient and controlled opening of the blood–brain barrier (BBB), a major obstacle for drug delivery to the brain.
The combination of ultrasound and microbubbles facilitates the targeted transport of therapeutic agents into brain tissue, particularly in the treatment of neurodegenerative diseases such as Alzheimer’s and Parkinson’s, as well as brain tumors.
This approach to ultrasound-mediated brain drug delivery opens new perspectives for non-invasive therapies.
🔹 Oncology
In oncology, sonoporation improves the penetration of anticancer agents into tumor tissues, which are often characterized by poor vascularization and limited drug diffusion.
By locally increasing membrane and tissue permeability, it promotes higher accumulation of therapeutic agents within the tumor, potentially enhancing treatment efficacy.
This approach also offers the possibility of reducing administered doses, thereby limiting the systemic side effects associated with chemotherapy.
🔹 Infectious Diseases
In the field of infectious diseases, sonoporation enhances the delivery of antiviral and antibacterial agents into hard-to-reach areas.
Certain infections persist due to the presence of biofilms or protective microenvironments that limit the effectiveness of conventional treatments.
Sonoporation improves the local diffusion of active compounds, particularly in poorly vascularized or chronically infected tissues.
🔹 Biomedical Research
In biomedical research, sonoporation is a valuable experimental tool for studying ultrasound–cell interactions.
It enables investigation of the relationship between mechanical forces and cellular responses, as well as membrane permeability mechanisms. It is also used to evaluate new drug delivery strategies in both in vitro and in vivo models.
Its flexibility and non-destructive nature make it a powerful technique for exploring innovative therapeutic approaches.
